Background: Sickle cell disease (SCD) is a lifelong, inherited disorder characterized by mutations in the hemoglobin (Hb) subunit β gene that leads to the production of sickle hemoglobin (HbS). When HbS is deoxygenated, polymerization leads to red blood cell sickling and damage, resulting in hemolysis, chronic anemia, and episodic vaso-occlusive crises (VOCs). Patients with SCD are at an increased risk of developing long-term complications, including stroke, leg ulcers, and other end-organ damage. Lower Hb levels highly correlate with increased morbidity and early mortality in SCD.
Voxelotor (Oxbryta®) is an oral, once-daily HbS polymerization inhibitor indicated for the treatment of SCD in adults and adolescent patients ≥12 years of age. The 24-week analysis of the HOPE trial demonstrated that treatment with voxelotor 1500 mg resulted in a significantly greater proportion of patients achieving a >1 g/dL Hb increase compared with placebo (51.1% vs 6.5%, P<0.001), which was associated with concordant improvements in hematological markers of hemolysis (indirect bilirubin and reticulocyte percentage). Here we report the evaluation of efficacy and safety of voxelotor 1500 mg at 72 weeks, the conclusion of the placebo-controlled HOPE trial.
Methods: In the randomized, double-blinded, placebo-controlled, phase 3 HOPE trial, adults and adolescents (aged 12-65 years) with SCD were randomized to receive voxelotor (1500 mg or 900 mg) or placebo. Included patients had an Hb level of 5.5 to 10.5 g/dL at enrollment and 1 to 10 vaso-occlusive crises in the 12 months prior to screening. Concurrent hydroxyurea was allowed if the dose had been stable for ≥90 days at enrollment. Changes from baseline in Hb and hemolysis markers (absolute and percentage reticulocyte, indirect bilirubin levels, and lactate dehydrogenase levels) and safety were assessed at week 72.
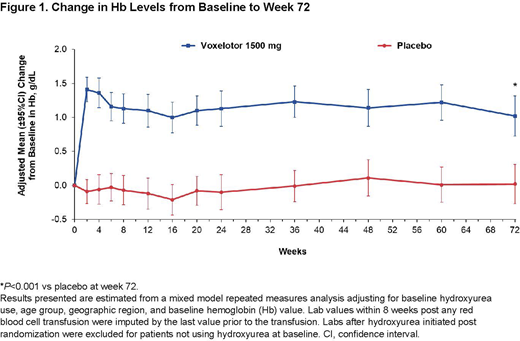
Results: 89% (95% CI, 82.4% to 95.4%) of patients receiving voxelotor 1500 mg achieved a Hb increase of >1 g/dL at 1 or more time points during the 72-week treatment period compared with 25% (95% CI, 16.2% to 33.8%) of those receiving placebo (P<0.001). The mean change in Hb from baseline at week 72 was 1.0 g/dL in patients treated with voxelotor 1500 mg compared with 0.0 g/dL in patients receiving placebo (Figure 1; P<0.001). Mean change from baseline to average Hb throughout the 72-week duration was 1.26 g/dL in patients treated with voxelotor 1500 mg. Consistent with the week 24 analysis, significant improvements in markers of hemolysis (assessed by difference in adjusted mean percent change versus placebo) were seen in indirect bilirubin (-26.6% [95% CI, -40.2% to -12.9%]) and reticulocyte percentage (-18.6% [95% CI, -33.9% to -3.3%]) in the voxelotor 1500 mg group relative to placebo, with favorable trends of reduction in other markers, such as absolute reticulocyte count (-5.8% [95% CI, -23.4% to 11.9%] and lactate dehydrogenase (-4.8% [95% CI, -13.8% to 4.1%]). The overall incidence rate of VOCs was numerically lower in the 1500 mg arm compared with placebo, but was not statistically significant. Rates of non-SCD and SCD-related treatment-emergent adverse events were similar between the treatment groups, with no new safety signals noted through week 72.
Conclusions: Voxelotor 1500 mg resulted in durable improvements in Hb levels and markers of hemolysis out to 72 weeks of treatment, with approximately 90% of patients achieving an increase in Hb >1 g/dL at 1 or more time points during the study. Treatment with voxelotor remained well tolerated, with no new safety signals detected with longer-term follow-up. These results support the sustained and chronic use of voxelotor to reduce anemia and hemolysis, thereby potentially mitigating the associated morbidity and mortality of SCD.
Howard:Imara, Inc., Novartis, Resonance Health: Honoraria; Agios, Forma Therapeutics, Inc., Global Blood Therapeutics, Imara, Inc., Novo Nordisk, Novartis: Membership on an entity's Board of Directors or advisory committees. Ataga:Editas Medicine: Honoraria; Global Blood Therapeutics: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Shire/Takeda: Research Funding; Novartis: Consultancy, Honoraria, Research Funding; Novo Nordisk: Honoraria, Membership on an entity's Board of Directors or advisory committees; Pfizer: Research Funding; Forma Therapeutics: Consultancy; Modus Therapeutics: Honoraria; Bioverativ: Honoraria, Membership on an entity's Board of Directors or advisory committees. Achebe:Global Blood Therapeutics: Consultancy. Hassab:Global Blood Therapeutics: Research Funding. Agodoa:Global Blood Therapeutics: Current Employment, Current equity holder in publicly-traded company. Tonda:Global Blood Therapeutics: Current Employment, Current equity holder in publicly-traded company. Gray:Global Blood Therapeutics: Current Employment, Current equity holder in publicly-traded company. Lehrer-Graiwer:Global Blood Therapeutics: Other: Former employee and former equity holder. Vichinsky:Pfizer: Research Funding; Agios: Research Funding; Global Blood Therapeutics: Consultancy.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal